Tirzepatide: What Dual Agonism Actually Does (2026 Research Guide)
Everything researchers need to know about tirzepatide's dual GIP/GLP-1 mechanism, SURMOUNT clinical trials, comparison to semaglutide and retatrutide, and sourcing for research.
Let me be crystal clear about something most tirzepatide guides bury: this compound didn't just beat semaglutide. It embarrassed it.
20.9% body weight loss at the highest dose. Nearly 21% in a population that couldn't crack 3% with lifestyle changes. Semaglutide's 14.9% looked incredible when STEP-1 dropped. Tirzepatide made it look like a warm-up.
And the mechanism isn't just "semaglutide plus GIP." The dual receptor activation creates metabolic effects that neither pathway achieves alone. Different fat metabolism. Different insulin sensitization. Different nausea profile. It's a genuinely different compound, not an incremental upgrade.
This guide covers everything: the dual mechanism, the SURMOUNT trial data, how tirzepatide stacks up against semaglutide and liraglutide, what's coming with retatrutide, and where to source it for legitimate research.
(No fluff. No hedging. Just the data.)
TL;DR
- Tirzepatide is a dual GIP/GLP-1 receptor agonist with 39 amino acids and a 5-day half-life via C20 fatty acid albumin binding
- Weight loss: 20.9% at 72 weeks (SURMOUNT-1, 15mg). Semaglutide reaches 14.9%, liraglutide 8%, retatrutide 24.2% (Phase 2)
- Dual advantage: GIP activation adds adipose lipid metabolism, peripheral insulin sensitization, and nausea attenuation that pure GLP-1 compounds can't deliver
- Head-to-head: SURMOUNT-5 showed 20.2% vs semaglutide's 13.7%, a 6.5 percentage point advantage (P<0.001)
- Pipeline context: Retatrutide adds a third receptor (glucagon) and may surpass tirzepatide, Phase 3 results expected 2026-27
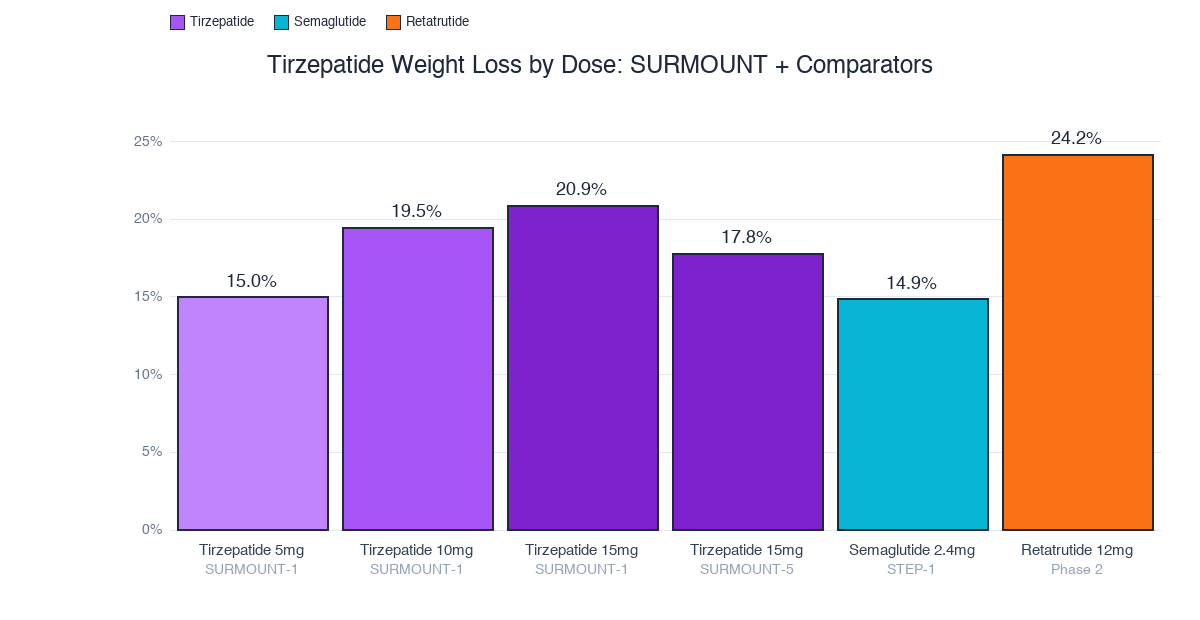
The dose-response curve is steep: 5mg delivers 15%, 15mg pushes to 20.9%. That 5mg-to-15mg jump is worth nearly 6 percentage points. Semaglutide maxes at 14.9% and retatrutide's triple agonism hits 24.2%.
What is Tirzepatide?
Tirzepatide is a lab-engineered peptide that activates two hormone receptors instead of one. It mimics both GIP (a gut hormone that helps process fat and sugar) and GLP-1 (the "I'm full" signal). The result is a compound that affects metabolism, appetite, and fat storage through two complementary pathways simultaneously.
Tirzepatide is a 39-amino acid synthetic dual agonist based on the native GIP sequence with structural modifications enabling both GIP and GLP-1 receptor activation.
The compound uses a GIP backbone (not GLP-1 like semaglutide) with specific modifications:
- Position 2 substitution: Aib at position 2 for DPP-4 resistance
- C20 fatty di-acid linker: Enables albumin binding for extended half-life
(This is the key structural difference from semaglutide. Semaglutide starts with a GLP-1 backbone and modifies it. Tirzepatide starts with a GIP backbone and engineers GLP-1 activity into it. Different starting points, different pharmacology.)
Molecular Identity:
| Property | Value |
|---|---|
| Molecular Weight | 4,813.53 g/mol |
| Amino Acids | 39 residues |
| CAS Number | 2023788-19-2 |
| PubChem CID | 156566512 |
| Receptor Targets | GIP + GLP-1 (dual) |
| Half-life | ~5 days (120 hours) |
| Protein Binding | >99% (albumin) |
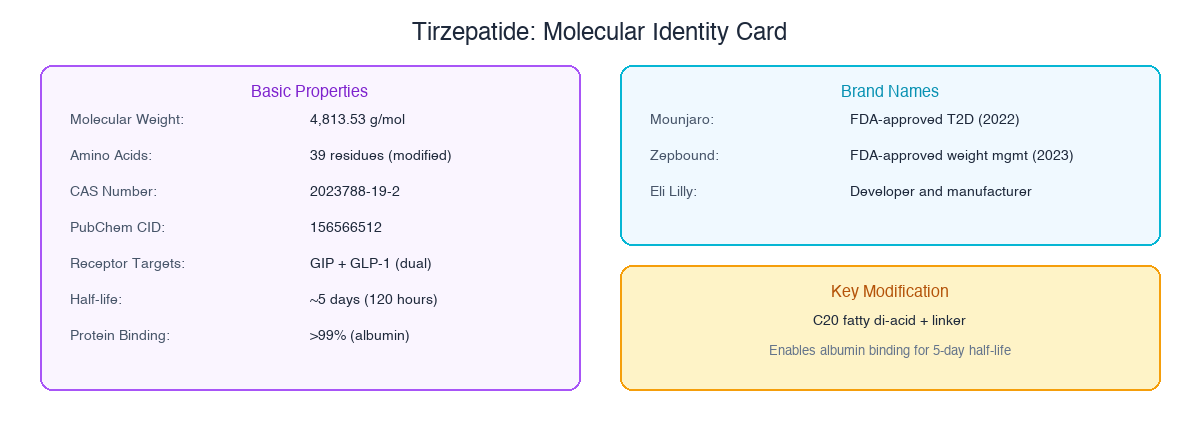
The two approved brands contain identical tirzepatide: Mounjaro for type 2 diabetes, Zepbound for weight management. Same molecule, same doses, different regulatory indication.
Pro Tip: If you're comparing tirzepatide to semaglutide at the molecular level, the backbone difference matters more than most people realize. GIP-based backbones interact with different intracellular trafficking pathways than GLP-1 backbones. This isn't just receptor selectivity, it's receptor manner. The same receptor can produce different downstream effects depending on how the agonist binds and internalizes.
How Tirzepatide Works
Tirzepatide works by hitting two receptors simultaneously. GLP-1 activation handles the appetite suppression and insulin secretion (same as semaglutide). GIP activation adds fat cell metabolism, bone protection, and reduces the nausea that GLP-1 alone causes. The two pathways reinforce each other.
Think of it like this: semaglutide is driving a car with one engine. Tirzepatide has two engines, and they share the same fuel system. Each engine does something slightly different, but they work better together than either one alone.
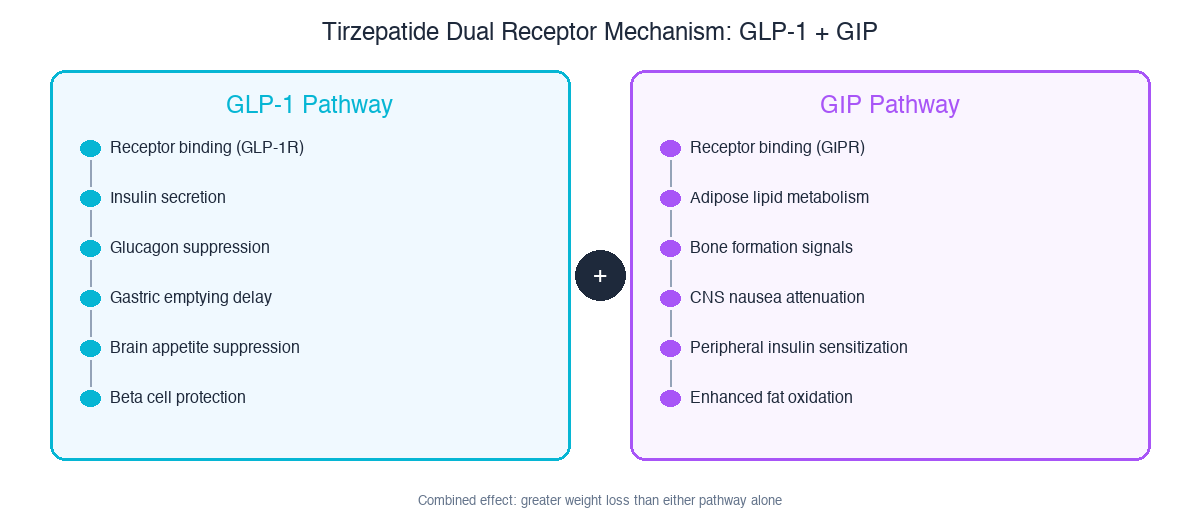
The GLP-1 pathway (left) handles the well-known effects: insulin secretion, glucagon suppression, gastric emptying, brain appetite control. The GIP pathway (right) adds what semaglutide can't do: adipose lipid metabolism, peripheral insulin sensitization, and nausea reduction.
The GLP-1 Component
Tirzepatide's GLP-1 receptor binding is 18-20 fold weaker than native GLP-1. This is intentional, not a weakness. The lower affinity with maintained GIP activity creates a synergistic effect:
- Insulin secretion: Glucose-dependent, like semaglutide
- Glucagon suppression: Reduces hepatic glucose output
- Gastric emptying: Delays absorption, improves satiety
- Brain signaling: Hypothalamic appetite suppression
(Semaglutide hits the GLP-1 receptor harder. Tirzepatide hits it smarter. The combination with GIP means you need less GLP-1 activation to get the same metabolic effect.)
The GIP Advantage (What Semaglutide Can't Do)
This is where tirzepatide pulls ahead. GIP receptors are expressed in tissues where GLP-1 receptors are absent or sparse:
Adipose tissue: GIP receptors on fat cells directly regulate lipoprotein lipase activity. This means tirzepatide improves how your body processes dietary fat, not just how it handles blood sugar. Semaglutide doesn't touch this pathway at all.
Nausea attenuation: GIP receptors in the area postrema (the brain's nausea center) partially counteract GLP-1-induced nausea. This is why tirzepatide often has slightly milder GI side effects than semaglutide despite being a more potent weight loss agent.
Peripheral insulin sensitization: The glucose disposal rate (M-value) improves beyond what weight loss alone predicts. This suggests direct insulin sensitization in muscle and liver tissue, mediated by GIP receptor activation.
Bottom line: The GIP component isn't just "bonus weight loss." It's a fundamentally different metabolic effect that opens research avenues pure GLP-1 compounds can't reach.
Safety Profile and Side Effects
Like any compound that actually works, tirzepatide comes with tradeoffs. The GI side effects are real but generally milder than semaglutide's, which is surprising given tirzepatide produces more weight loss. The serious risks are rare but follow the same pattern as other incretin agonists.
Common Side Effects
| Side Effect | Tirzepatide | Semaglutide | Placebo |
|---|---|---|---|
| Nausea | 18% | 44% | 16% |
| Diarrhea | 12% | 30% | 16% |
| Vomiting | 6% | 25% | 6% |
| Constipation | 8% | 24% | 11% |
| Appetite Decrease | 22% | 35% | 5% |
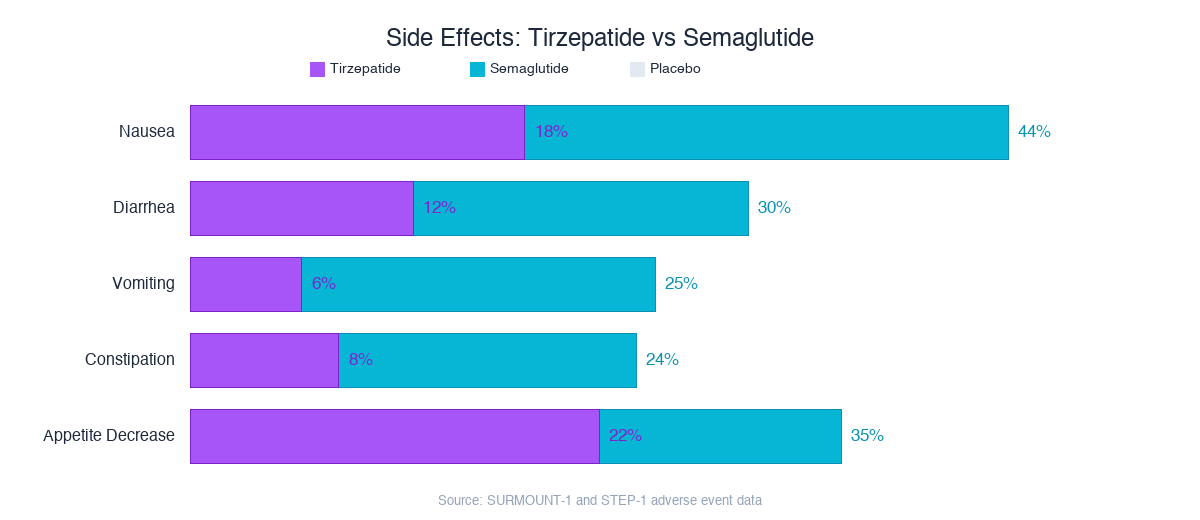
The nausea gap is striking: tirzepatide at 18% vs semaglutide at 44%. The GIP-mediated nausea attenuation in the area postrema appears to genuinely reduce GI distress despite comparable or superior weight loss efficacy.
(Liraglutide sits between the two, with nausea rates around 20-25%. The pattern is clear: more GLP-1 receptor activation means more nausea, and GIP co-activation provides a buffer.)
Serious Adverse Events
Pancreatitis: 0.2% incidence in clinical trials. Monitor amylase and lipase levels in animal studies. Same mechanism as semaglutide, direct stimulation of pancreatic acinar cells.
Gallbladder disease: 1.5% incidence, driven primarily by rapid weight loss rather than direct drug effect. Higher rates at faster weight loss speeds.
Thyroid C-cell tumors: Boxed warning from rodent studies at supratherapeutic doses. Human relevance unconfirmed. Contraindicated with personal/family history of medullary thyroid carcinoma or MEN2 syndrome.
Injection site reactions: 3-5% incidence, mild (erythema, pruritus). Rotation of injection sites minimizes occurrence.
The Clinical Trial Data That Actually Matters
The SURMOUNT program tested tirzepatide in over 6,000 people across 5 Phase 3 trials. The results are consistent: 15-21% weight loss depending on dose and population, superior to semaglutide head-to-head, with metabolic benefits extending beyond the scale.
Let me be crystal clear: tirzepatide's clinical dataset is robust. Not as deep as semaglutide's 80,000+ participants, but the signal is stronger per trial. The dose-response relationship is steep and the head-to-head data is definitive.
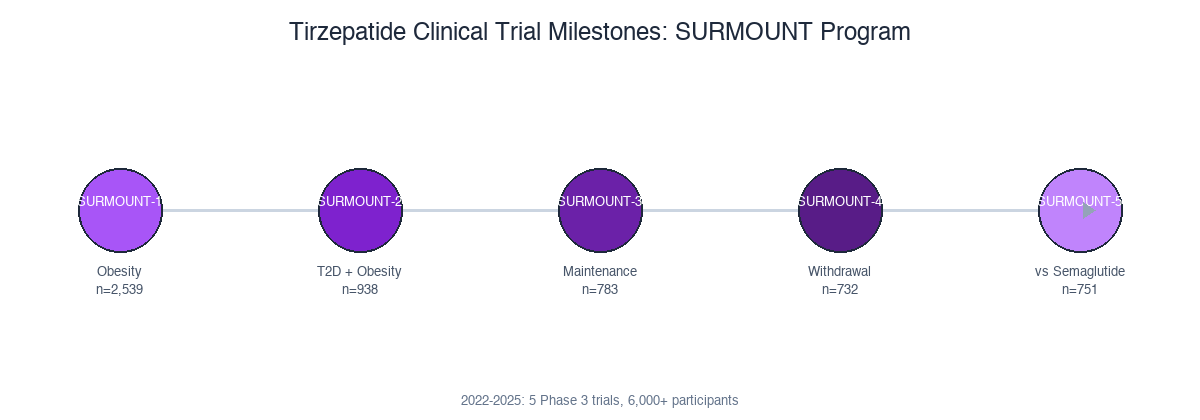
Each SURMOUNT trial tested a different question: efficacy in obesity, efficacy in T2D, maintenance after weight loss, weight regain after withdrawal, and head-to-head against semaglutide. Together they paint a complete picture.
SURMOUNT-1: Obesity Without Diabetes (n=2,539)
| Dose | Mean Weight Loss | Participants Achieving >=5% Loss |
|---|---|---|
| 5mg | 15.0% | 85% |
| 10mg | 19.5% | 89% |
| 15mg | 20.9% | 91% |
| Placebo | 3.1% | 35% |
Source: Jastreboff et al., N Engl J Med 2022;387:205-216
Read that 15mg number again. 20.9% body weight loss. For context, bariatric surgery typically achieves 25-30% at one year. Tirzepatide is approaching surgical results without an operation.
(Semaglutide's STEP-1 produced 14.9%. Tirzepatide beat it by 6 percentage points in comparable populations. That's not a marginal improvement, it's a different tier.)
SURMOUNT-2: Obesity With Type 2 Diabetes (n=938)
| Dose | Mean Weight Loss | HbA1c Reduction |
|---|---|---|
| 10mg | 12.8% | 2.1% |
| 15mg | 14.7% | 2.1% |
71% of participants on 10mg and 83% on 15mg achieved >=5% weight loss despite baseline diabetes. The weight loss is attenuated vs non-diabetic populations (similar pattern seen with semaglutide in STEP-2), but the HbA1c reduction is exceptional.
SURMOUNT-5: Head-to-Head vs Semaglutide (n=751)

The 6.5 percentage point gap was statistically significant at P<0.001. Tirzepatide also showed greater waist circumference reduction (-18.4cm vs -13.0cm), suggesting more visceral fat loss.
| Parameter | Tirzepatide (10/15mg) | Semaglutide (1.7/2.4mg) | Difference |
|---|---|---|---|
| Mean weight loss | 20.2% | 13.7% | +6.5% tirzepatide |
| Waist reduction | -18.4cm | -13.0cm | +5.4cm tirzepatide |
Source: Aronne et al., N Engl J Med 2025;393:26-36
Bottom line: The head-to-head data is definitive. Tirzepatide produces more weight loss, more waist reduction, and more metabolic improvement than semaglutide. The GIP component adds a measurable advantage.
Cardiovascular and Renal Effects
SUMMIT trial (HFpEF): Tirzepatide produced a 38% reduction in cardiovascular death or worsening heart failure (HR 0.62) in participants with obesity and preserved ejection fraction.
Renal protection: Analysis across SURPASS and SURMOUNT showed slower eGFR decline and reduced UACR, with 54% of the UACR reduction independent of weight or HbA1c changes.
(Liraglutide's LEADER trial showed CV benefit in diabetics. Semaglutide's SELECT showed it in non-diabetics. Tirzepatide's SUMMIT adds heart failure to the list. The incretin class keeps expanding its cardiovascular evidence base.)
Head-to-Head: Tirzepatide vs Semaglutide vs Liraglutide
Tirzepatide vs Semaglutide
The SURMOUNT-5 data settled this debate:
- Weight loss: Tirzepatide wins by 6.5 percentage points
- Metabolic markers: Greater improvements in triglycerides, apoB, blood pressure
- Safety profile: Tirzepatide has milder GI side effects despite superior efficacy
- Long-term data: Semaglutide has 5+ more years of safety data and broader approved indications
For researchers: If you're studying incretin synergy, tirzepatide is the compound. If you need the deepest safety dataset or pure GLP-1 mechanism, semaglutide remains the cleaner tool. Both are valid research choices depending on the question.
Choose tirzepatide when: Maximal weight loss is the goal, studying dual agonism mechanisms, or investigating GIP-specific adipose effects. Also when researching switch protocols from semaglutide (60-70% of plateau-breakers respond to the switch).
Choose semaglutide when: Pure GLP-1 mechanism is the research question, budget constraints exist, or cardiovascular/MASH data is the primary focus (semaglutide has broader indication approval).
Tirzepatide vs Liraglutide
| Feature | Tirzepatide | Liraglutide |
|---|---|---|
| Receptor targets | GIP + GLP-1 | GLP-1 only |
| Half-life | ~5 days (weekly) | ~13 hours (daily) |
| Weight loss | 15-21% | 8-10% |
| HbA1c reduction | 2.1% | 1.0-1.5% |
| Nausea rate | 18% | 20-25% |
Liraglutide proved daily GLP-1 agonism could work. Semaglutide proved weekly dosing was better. Tirzepatide proved adding a second receptor doubles the effect. The lineage is clear: single target, longer half-life, dual target, triple target (retatrutide).
The Pipeline: Retatrutide
Retatrutide adds a third receptor to the equation: glucagon receptor activation alongside GIP and GLP-1.
Phase 2 data (48 weeks):
| Dose | Weight Loss |
|---|---|
| 1mg | 8.7% |
| 4mg | 16.9% |
| 8mg | 22.8% |
| 12mg | 24.2% |
Read that 12mg number again. 24.2% in 48 weeks. Tirzepatide needs 72 weeks to reach 20.9%. Retatrutide beats it in less time.
What glucagon receptor activation adds: Counterintuitively, activating a receptor that raises blood sugar appears to increase energy expenditure and fat oxidation when combined with GIP and GLP-1 agonism. The triple synergy appears to be greater than the sum of its parts.
Phase 3 trials are ongoing. If confirmed, retatrutide becomes the new gold standard and tirzepatide becomes the "proven but not best" option.
For researchers building programs now: Design protocols that can accommodate retatrutide data. The GIP and GLP-1 receptor binding assays you'd run for tirzepatide translate directly, you'd just need additional glucagon receptor readouts.
Research Applications
The dual receptor mechanism opens research doors that single-pathway compounds can't. Five main fields are actively investigating tirzepatide, from established metabolic studies to emerging comparative research.
Current Laboratory Studies
Metabolic Research:
- Insulin secretion dynamics (dual pathway analysis)
- Beta cell preservation studies
- Glucose homeostasis mechanism investigations
- Adipose lipid metabolism (GIP-specific)
Cardiovascular Biology:
- HFpEF outcomes and cardiac metabolism
- Lipid optimization (apoB reduction mechanisms)
- Endothelial function and atherosclerosis models
- MACE reduction pathway studies
Renal Research:
- eGFR preservation mechanisms
- UACR reduction (54% independent of weight/HbA1c)
- Glomerular protection pathways
- Direct renoprotective effects
Hepatology:
- MASH/NASH treatment potential
- Hepatic steatosis improvement
- Fibrosis regression mechanisms
Comparative Studies:
- Tirzepatide vs semaglutide mechanistic differences
- GIP-specific vs GLP-1-only pathway mapping
- Dual vs triple agonism (with retatrutide)
- Switch protocol research (semaglutide to tirzepatide)
Pro Tip: If you're studying adipose tissue biology specifically, tirzepatide is the compound of choice. The GIP receptor on adipocytes regulates lipoprotein lipase activity directly. Semaglutide doesn't touch this pathway. This is a genuine research differentiator.
In-Vitro Protocol Reference
Receptor Binding Assays:
- GIPR binding: CHO cells expressing human GIP receptor, IC50 ~0.1 nM
- GLP1R binding: CHO cells expressing human GLP-1 receptor, IC50 ~1.0 nM (weaker than semaglutide)
- Dual readout: Set up parallel plates for both receptors
cAMP Accumulation:
- Cell lines: INS-1, MIN6, CHO-GIPR, CHO-GLP1R
- EC50 varies by receptor: ~0.05 nM for GIPR, ~0.3 nM for GLP1R
Pro Tip: When comparing tirzepatide against semaglutide in binding assays, the GLP1R binding affinity difference is the key differentiator. Tirzepatide's GLP1R Ki is 18-20x weaker, but its overall metabolic effect is stronger because of the GIP component. Don't misinterpret weaker receptor binding as weaker compound efficacy.
Sourcing for Research
If you're buying tirzepatide for research, the dual agonism creates an additional verification requirement: you need to confirm BOTH receptor activities, not just one. A compound that only activates GLP-1 is not tirzepatide, it's semaglutide with a wrong label.
When sourcing tirzepatide for laboratory applications, researchers should prioritize quality verification and vendor reliability.
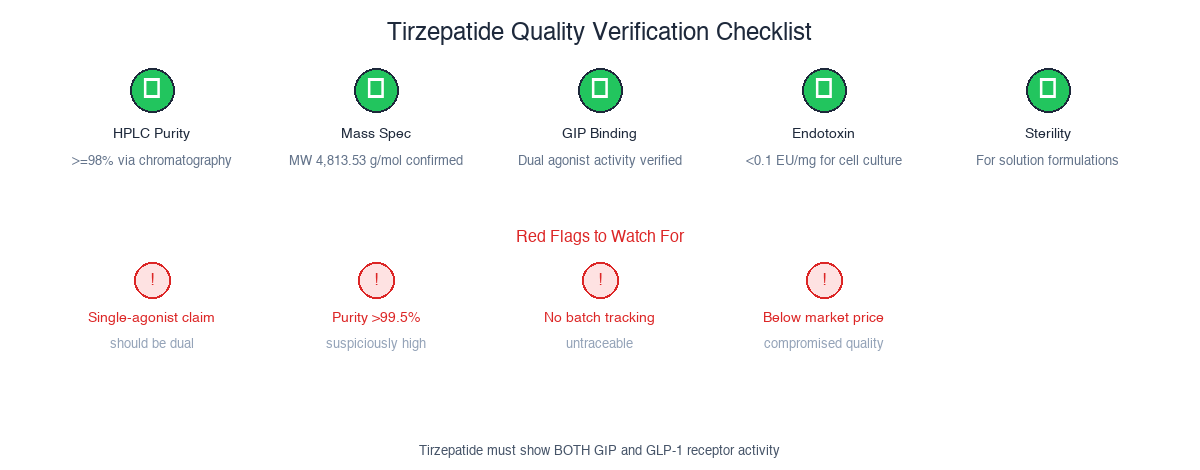
The five non-negotiable checks for research-grade tirzepatide. The critical difference from semaglutide verification: you must confirm dual agonist activity, not just purity. A pure compound that only hits GLP-1 receptors is not tirzepatide.
Quality Verification Checklist
Before procurement, verify:
- HPLC Purity: >=98% via high-performance liquid chromatography
- Mass Spectrometry: Molecular weight confirmation (4,813.53 g/mol)
- Dual Receptor Binding: GIP and GLP-1 receptor activity confirmed
- Peptide Content: Amino acid analysis for 39-residue sequence
- Endotoxin Levels: <0.1 EU/mg for cell culture applications
Storage and Handling
Lyophilized Peptide:
- Storage: -20C (short-term), -80C (long-term)
- Stability: 2-3 years properly stored
- Protect from light and moisture
Reconstituted Solution:
- Use sterile bacteriostatic water
- Storage: 2-8C for 28 days
- Avoid freeze-thaw cycles
- Typical concentration: 2-10 mg/mL
Where to Buy Tirzepatide for Research
Featured verified vendors that list tirzepatide in the directory (up to five, randomized per visit; availability can change):
For the full screened list, see the verified vendor directory.
Conclusion
Tirzepatide proved that dual incretin agonism produces superior metabolic effects compared to single-pathway compounds. The GIP component isn't just additive, it's synergistic. It adds fat metabolism, reduces nausea, and delivers 6+ percentage points more weight loss than semaglutide.
For researchers, tirzepatide offers a unique tool: a single molecule that simultaneously activates two receptor pathways with distinct downstream effects. This opens mechanistic studies that pure GLP-1 compounds can't support.
But here's the bottom line: liraglutide proved daily GLP-1 could work. Semaglutide proved weekly GLP-1 worked better. Tirzepatide proved adding GIP doubles the effect. Retatrutide is testing whether adding glucagon completes the picture. The incretin story keeps getting more complex and more effective with each iteration. Tirzepatide is the current chapter, not the final one.
Where to Go from Here
If you're starting a dual agonist research program:
- Begin with tirzepatide receptor binding assays (both GIPR and GLP1R)
- Add semaglutide as a GLP-1-only comparator
- Design protocols with retatrutide compatibility for the next phase
- Source from verified vendors with dual binding confirmation
If you're expanding an existing program:
- The adipose tissue angle is GIP-specific and underserved
- Switch protocols (semaglutide to tirzepatide) have 60-70% plateau-breaking response
- Heart failure (SUMMIT) opens cardiovascular research beyond traditional MACE endpoints
For researchers seeking to incorporate this compound into laboratory protocols, explore our verified vendor directory or browse related peptides in our fat loss research category.
Research Notice: This guide is provided for educational and research purposes only. BestPeptide.info does not condone the misuse of research chemicals. Always consult qualified professionals regarding regulatory compliance, safety protocols, and institutional review board requirements.
References: Key studies cited include Jastreboff et al. (2022) SURMOUNT-1 in NEJM, Aronne et al. (2025) SURMOUNT-5 in NEJM, and Kosiborod et al. (2024) SUMMIT in NEJM. Full citations available upon request.