Semaglutide: What the Clinical Data Actually Shows (2026 Research Guide)
Everything researchers need to know about semaglutide, mechanism, clinical trials (SUSTAIN, STEP, SELECT), sourcing, and what's coming next including retatrutide.
Here's the thing about semaglutide that most guides won't tell you upfront: it's not the final boss of GLP-1 research anymore.
Sure, semaglutide rewrote the rules. 15% body weight loss in a clinical population that couldn't crack 3% with lifestyle changes alone. A 20% reduction in cardiovascular events in non-diabetics. Over 80,000 trial participants across three massive programs.
The data is overwhelming. And the research applications keep expanding: diabetes, obesity, cardiovascular protection, liver disease, and now possibly Alzheimer's. Semaglutide isn't just a weight loss drug. It's become the Rosetta Stone for understanding GLP-1 receptor biology at scale.
But there's a newer compound lurking in Phase 3 trials right now, and if you're building a research program around GLP-1 biology, you need to understand both.
This guide covers everything: the molecular mechanics, the clinical trial data that actually matters, how semaglutide stacks up against tirzepatide and liraglutide, and what's coming next with retatrutide. Plus, where to source it for legitimate research and how to verify you're getting the real thing.
(No fluff. No hedging. Just the data.)
TL;DR
- Semaglutide is a modified GLP-1 analog with 94% homology to human GLP-1, extended to a 7-day half-life via albumin-binding acylation
- Weight loss: 14.9% at 68 weeks (STEP-1), with 69% of participants losing 10% or more. Liraglutide manages 8-10%, tirzepatide reaches 17.8% (SURMOUNT-5)
- Cardiovascular: 20% MACE reduction in the SELECT trial (n=17,604), first proven CV benefit in non-diabetics
- Receptor: Selective GLP-1R agonist. Tirzepatide adds GIP. Retatrutide adds GIP + glucagon. Semaglutide remains the cleanest pure GLP-1 tool
- Pipeline threat: Retatrutide (triple agonist) showed 24% weight loss in Phase 2, Phase 3 results expected 2026-27. Tirzepatide already outperforms semaglutide head-to-head (SURMOUNT-5)
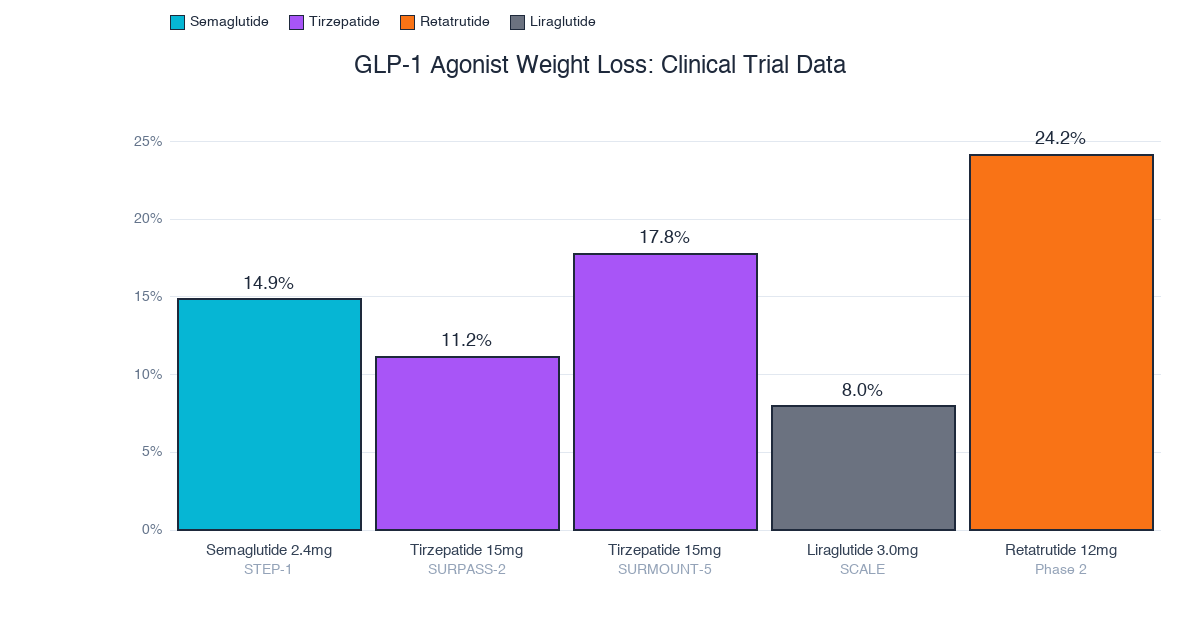
The weight loss hierarchy is clear: retatrutide's triple agonism (24.2%) outperforms everything, tirzepatide's dual agonism (17.8%) beats semaglutide's pure GLP-1 approach (14.9%), and liraglutide trails at 8.0%. But more receptors means more unknowns, semaglutide has the deepest safety dataset.
What is Semaglutide?
Semaglutide is essentially a synthetic copy of a hormone your body already makes. Scientists modified it slightly so it lasts a full week instead of minutes. This extended effect means once-weekly dosing instead of multiple daily injections.
Semaglutide is a synthetic analog of human glucagon-like peptide-1 (GLP-1) consisting of a 31-amino acid peptide backbone with specific structural modifications designed to extend half-life and enhance receptor binding.
You might know it by its brand names: Ozempic (diabetes, injectable), Wegovy (weight management, injectable), and Rybelsus (diabetes, oral tablet). Same molecule, different packaging, different approved indications. Novo Nordisk developed all three.
The injectable vs oral distinction matters for researchers. The subcutaneous injection (Ozempic/Wegovy) delivers the full peptide with ~97% bioavailability. The oral tablet (Rybelsus) uses an absorption enhancer (SNAC, sodium salcaprozate) to protect the peptide through stomach acid and enable limited intestinal absorption, but bioavailability drops to about 1%. That's why the oral dose is 14mg daily vs the weekly injection at 0.25-2.4mg.
Here's what makes it different from the native hormone your gut produces after every meal:
The native GLP-1 sequence (7-37) serves as the foundation. Two structural changes turn a hormone that lasts minutes into one that lasts a full week:
- Position 8 Substitution: Alpha-aminoisobutyric acid (Aib) replaces alanine, protecting against dipeptidyl peptidase-4 (DPP-4) degradation (this is what keeps your body from shredding it immediately)
- Position 26 Acylation: A C-18 fatty di-acid (octadecanedioic acid) attached via a gamma-Glu linker enables non-covalent albumin binding (and this is what keeps it circulating for days instead of minutes)
The albumin binding from that C-18 fatty acid modification is the real magic. It reduces renal clearance and extends circulating half-life from minutes (native GLP-1) to 168 hours, enabling weekly administration protocols in research settings.
Molecular Properties:
| Property | Value |
|---|---|
| PubChem CID | 56843331 |
| Molecular Formula | C187H291N45O59 |
| Molecular Weight | 4,114 g/mol |
| CAS Number | 910463-68-2 |
| Sequence Homology | 94% identical to human GLP-1 |
| Receptor Binding (Ki) | 0.38 +/- 0.06 nM |
| Plasma Half-life | 168 hours (7 days) |
| Protein Binding | >99% (albumin) |
Source: PubChem CID 56843331
Pro Tip: If you're comparing semaglutide to other GLP-1 analogs, the albumin binding mechanism is the differentiator. Liraglutide uses a C-16 fatty acid (shorter chain, less binding affinity). That's why liraglutide lasts 13 hours and semaglutide lasts 7 days. Same concept, better execution. Tirzepatide took a different path entirely, adding a GIP backbone with its own fatty acid linker. If you're studying the pharmacokinetic structure-activity relationship, comparing all three side by side is incredibly revealing.
How Semaglutide Works
Semaglutide works in four main ways: (1) It tells your brain you're full, reducing appetite. (2) It slows stomach emptying, so food satisfies you longer. (3) It triggers insulin release when blood sugar rises. (4) It reduces sugar production by the liver. The combined effect: you eat less, feel satisfied with smaller portions, and your metabolism handles sugar better.
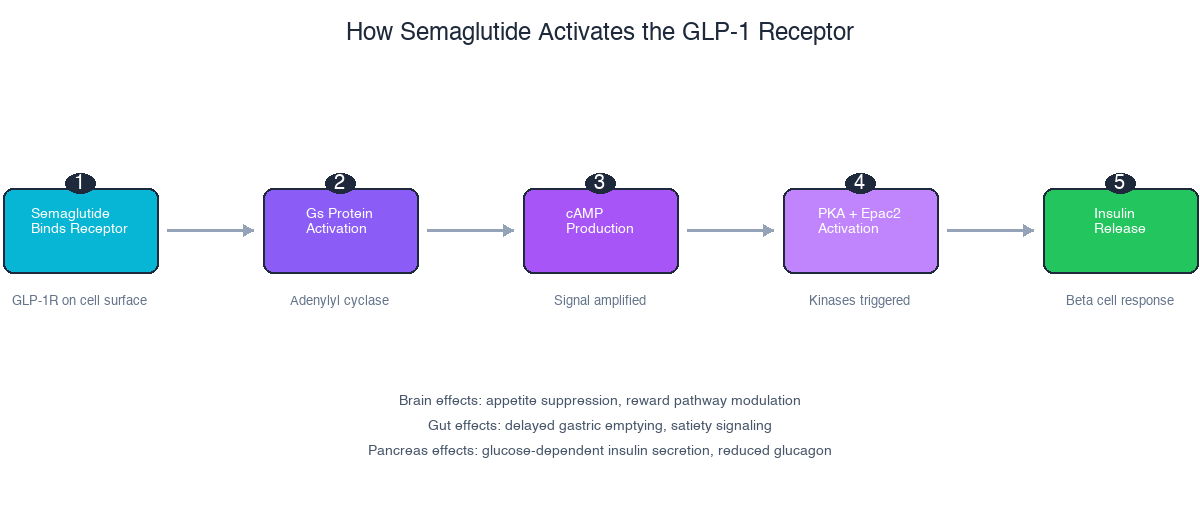
The diagram shows the five-step signaling cascade: semaglutide binds the GLP-1 receptor, triggering Gs protein activation, cAMP production, PKA/Epac2 kinase activation, and ultimately insulin release from pancreatic beta cells. The same receptor activation also produces effects in the brain (appetite suppression) and gut (delayed gastric emptying).
Semaglutide functions as a potent, selective agonist at the GLP-1 receptor, a class B G-protein coupled receptor expressed in pancreatic beta cells, gastrointestinal tract, brainstem nuclei, and hypothalamic regions.
Think of it like this: semaglutide is hitting the same receptor that your body's natural GLP-1 hits after a meal. But because it sticks around for a week instead of minutes, it's like having that "I just ate" signal running 24/7.
Why selectivity matters for researchers: Liraglutide also hits the GLP-1 receptor selectively, but its shorter half-life means the receptor occupancy profile is completely different. Tirzepatide hits GLP-1R and GIP receptor, which creates crosstalk effects that complicate mechanistic studies. If your research question is specifically about GLP-1 receptor biology, semaglutide gives you the cleanest signal without the confound of GIP receptor activation.
The Signaling Cascade
- Semaglutide binds to the extracellular domain of the GLP-1 receptor
- Induces conformational change activating the transmembrane domain
- Stimulates Gs-protein coupling, increasing intracellular cAMP
- Activates protein kinase A (PKA) and Epac2 pathways
- Modulates ion channel activity and gene expression
What happens downstream in research models:
- Calcium Influx: Enhanced intracellular calcium in beta cells
- Insulin Gene Transcription: Upregulation via PDX-1 and MafA activation
- Beta Cell Proliferation: Promotion of cellular replication (in vitro evidence)
- Anti-apoptotic Effects: Reduced beta cell death under stress conditions
Four Physiological Mechanisms
1. Glucose-Dependent Insulin Secretion
Semaglutide amplifies glucose-stimulated insulin release through direct enhancement of insulin granule exocytosis, increased beta cell sensitivity to glucose, preservation of first-phase insulin response, and reduction of glucagon secretion.
Bottom line: it only pushes insulin when blood sugar is actually high. That's why hypoglycemia risk is low.
2. Gastrointestinal Effects
The compound delays gastric emptying, slowing nutrient absorption and reducing postprandial glucose excursions. It also modifies small bowel transit and activates vagal afferent pathways contributing to satiety signaling.
This is the mechanism behind the "I can barely finish a plate" experience that trial participants consistently report.
3. Central Nervous System Actions
Semaglutide crosses the blood-brain barrier at low concentrations, activating GLP-1 receptors in multiple brain regions:
Hypothalamic nuclei:
- Arcuate nucleus: Contains POMC/CART neurons that suppress appetite and NPY/AgRP neurons that promote hunger. Semaglutide activates the satiety neurons and inhibits the hunger neurons simultaneously.
- Paraventricular nucleus: Integrates energy balance signals and controls downstream thyroid and adrenal axes.
- Dorsomedial hypothalamus: Involved in circadian feeding patterns and thermoregulation.
Brainstem regions:
- Nucleus tractus solitarius (NTS): Primary relay for gut-derived satiety signals. Semaglutide amplifies the vagal afferent input here.
- Area postrema: Circumventricular organ outside the blood-brain barrier. Directly exposed to circulating semaglutide, contributing to the nausea side effect (this is actually the mechanism, not a bug).
Reward pathways:
- Ventral tegmental area (VTA): Dopaminergic neurons involved in food reward processing.
- Nucleus accumbens: Pleasure/reward center. Semaglutide appears to reduce the hedonic drive to eat high-palatability foods.
The reward pathway activation is underappreciated. Early research suggests semaglutide may reduce the hedonic drive to eat, not just the homeostatic hunger signal. This is why participants report reduced cravings for specific foods, not just reduced appetite generally.
Researchers studying addiction have taken notice. Preliminary data suggests GLP-1R agonism may reduce alcohol consumption, nicotine cravings, and even opioid-seeking behavior. The mechanism likely involves dampening dopaminergic reward signaling in the mesolimbic pathway.
4. Hepatic Effects
Reduction of hepatic glucose production and improvement in insulin sensitivity at the liver level. This contributes to fasting glucose improvement independent of weight loss.
The MASH/ESSENCE approval in 2025 was built on this mechanism. Semaglutide reduced liver fibrosis and steatohepatitis in clinical trials, making it the first GLP-1 agonist approved for liver disease.
Safety Profile and Side Effects
Like any medication that actually works, semaglutide comes with tradeoffs. The most common side effects are nausea, vomiting, and diarrhea, especially when starting or increasing doses. These usually improve over time as your body adjusts. The serious risks are rare but real: pancreatitis, gallbladder problems, and a thyroid cancer signal in rodents (not confirmed in humans).
Let me be direct: if you're researching semaglutide, you need to understand both sides of the risk-benefit equation.
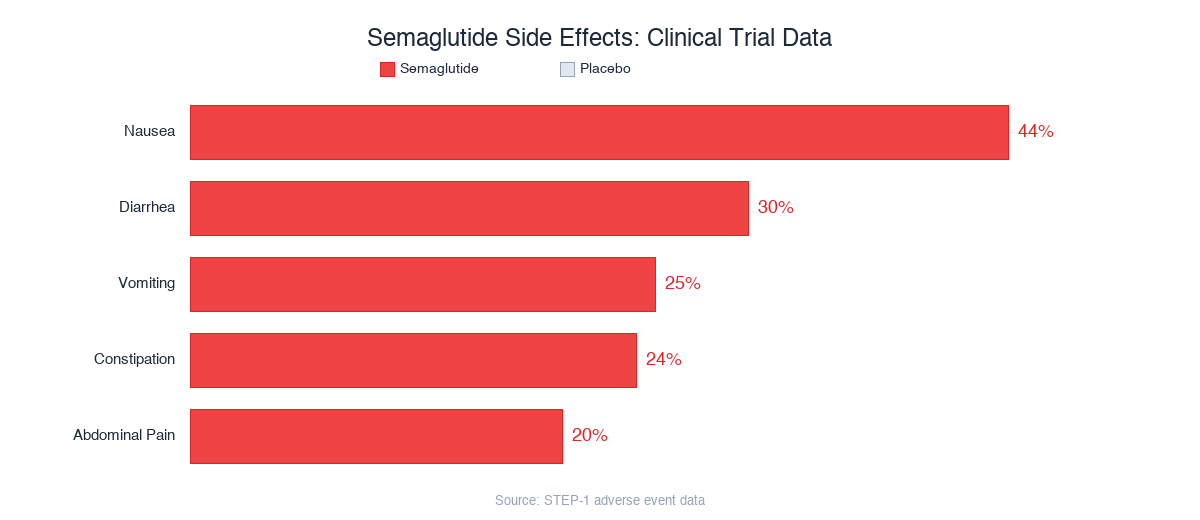
Nausea is by far the most common side effect, hitting nearly half of participants in STEP-1. But notice the placebo rates too, gut issues are common in this population regardless. The dose escalation schedule (starting at 0.25mg and slowly increasing) exists specifically because starting at full dose causes severe GI effects.
Common Side Effects (Phase 3 Data)
| Side Effect | Semaglutide | Placebo |
|---|---|---|
| Nausea | 44% | 16% |
| Vomiting | 25% | 6% |
| Diarrhea | 30% | 16% |
| Constipation | 24% | 11% |
| Abdominal pain | 20% | 9% |
| Injection site reactions | 1% | 1% |
Source: STEP-1 adverse event data
The nausea pattern is predictable and manageable in research protocols. Clinical trials use a gradual dose escalation (0.25mg to 0.5mg to 1mg to 1.7mg to 2.4mg over 16-20 weeks) specifically because starting at full dose causes severe GI effects in most participants. Any research protocol should mirror this titration schedule.
(Worth noting: liraglutide has a similar GI side effect profile but slightly lower rates overall, likely due to lower peak receptor occupancy with daily dosing. Tirzepatide's nausea rates are comparable to semaglutide despite the added GIP activity, suggesting the GI effects are primarily GLP-1R mediated.)
Serious Adverse Events
Pancreatitis: Incidence of 0.2-0.4% in clinical trials (vs 0.1% placebo). Researchers should monitor amylase and lipase levels in animal studies. The mechanism involves direct stimulation of pancreatic acinar cells.
Gallbladder disease: Increased risk of cholelithiasis (gallstones), likely due to rapid weight loss rather than direct drug effect. Incidence ~2.5% vs 1.2% placebo in STEP trials.
Thyroid C signal: Semaglutide caused thyroid C-cell tumors in rodent studies at supratherapeutic doses. Human relevance is unconfirmed, but the drug carries a boxed warning against use in patients with personal/family history of medullary thyroid carcinoma or MEN2 syndrome. (This is a precautionary label, not a confirmed human risk, but researchers should note it.)
Diabetic retinopathy complications: SUSTAIN-6 showed a small increase in diabetic retinopathy events (3.0% vs 1.8%). The mechanism likely involves rapid glucose normalization causing temporary retinal vascular changes, not direct toxicity.
Drug Interactions to Note
Semaglutide's gastric emptying delay affects oral medication absorption. Key interactions:
- Insulin and sulfonylureas: Increased hypoglycemia risk (dose reduction often needed)
- Oral medications: Delayed absorption due to slowed gastric emptying
- Levothyroxine: May require dose adjustment due to absorption changes
For researchers: If you're running in-vivo studies with combination protocols, account for the gastric emptying effect. Oral drugs administered within 1 hour of semaglutide injection may show altered pharmacokinetics.
The Clinical Trial Data That Actually Matters
Semaglutide has been tested in over 80,000 people across three major programs: SUSTAIN (for diabetes), STEP (for weight loss), and SELECT (for heart protection). The results are remarkably consistent, substantial weight loss, better blood sugar control, and reduced cardiovascular risk. This isn't based on one small study; it's one of the most thoroughly tested medications in existence.
Let me be crystal clear about something: the clinical dataset behind semaglutide is massive. We're not talking about a Phase 2 trial with 200 people. We're talking about 80,000+ participants across multiple Phase 3 programs with consistent results.
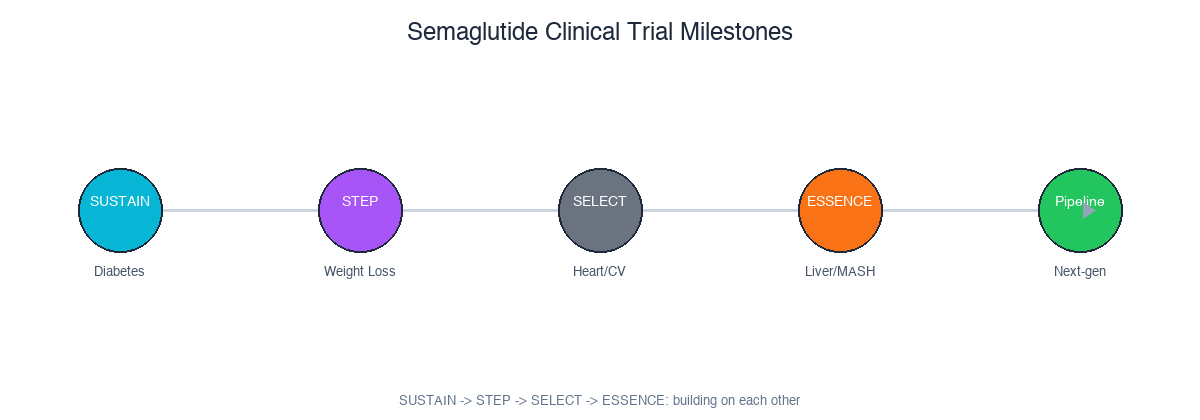
Each trial program built on the last: SUSTAIN proved diabetes efficacy, STEP proved weight loss, SELECT proved cardiovascular benefit in non-diabetics, and ESSENCE secured MASH approval. The pipeline represents the next frontier.
SUSTAIN Program (Type 2 Diabetes)
The SUSTAIN (Semaglutide Unabated Sustainability in Treatment of Type 2 Diabetes) program established semaglutide as a superior diabetes treatment across multiple comparator arms:
| Trial | Population | Duration | Participants | Key Finding |
|---|---|---|---|---|
| SUSTAIN 1 | Drug-naive T2D | 30 weeks | 388 | HbA1c -1.5% vs placebo |
| SUSTAIN 2 | Metformin failures | 56 weeks | 1,231 | Superior to sitagliptin (-1.1% vs -0.6%) |
| SUSTAIN 3 | Metformin +/- sulfonylurea | 56 weeks | 1,204 | Superior to exenatide ER (-1.21% vs -0.99%) |
| SUSTAIN 4 | Metformin +/- sulfonylurea | 30 weeks | 1,089 | Superior to insulin glargine (-1.21% vs -0.54%) |
| SUSTAIN 5 | Insulin-treated T2D | 30 weeks | 397 | Add-on to insulin: HbA1c -1.41% |
| SUSTAIN 6 | High CV risk T2D | 104 weeks | 3,297 | 26% MACE reduction (p=0.02) |
Source: Marso et al., N Engl J Med 2016;374:1834-1844 (SUSTAIN-6)
What SUSTAIN 6 proved: Semaglutide didn't just lower blood sugar. It reduced major cardiovascular events by 26% in high-risk diabetics. That's the trial that changed the conversation from "diabetes drug" to "cardiovascular protective agent."
(Liraglutide had shown similar CV benefit in LEADER back in 2016, but with a smaller effect size and only in diabetics. Semaglutide's SELECT trial later proved it works in non-diabetics too, which liraglutide never demonstrated.)
STEP Program (Weight Management)
This is where semaglutide became a household name. The STEP program evaluated semaglutide 2.4mg for weight management:
| Trial | Population | Duration | Participants | Weight Loss | >=10% Loss |
|---|---|---|---|---|---|
| STEP 1 | Obesity/Overweight | 68 weeks | 1,961 | 14.9% | 69% |
| STEP 2 | T2D + Overweight | 68 weeks | 1,210 | 9.6% | 51% |
| STEP 3 | Obesity + IBT | 68 weeks | 611 | 16.0% | 76% |
| STEP 4 | Withdrawal study | 68+52 weeks | 902 | Rebound +11.6% | N/A |
| STEP 5 | Obesity | 104 weeks | 304 | 15.2% maintained | 67% |
Source: Wilding et al., N Engl J Med 2021;384:989-1002 (STEP-1)
Read that STEP 1 number again. 14.9% body weight loss. For context, the previous best anti-obesity drug (liraglutide at 3.0mg) managed 8-10%. Lifestyle interventions alone typically produce 2-3%. This was a paradigm shift.
(Tirzepatide would later push this to 17.8% in SURMOUNT-5, but STEP-1 was the trial that proved GLP-1 agonism alone could deliver transformational weight loss. Semaglutide opened the door.)
The STEP 4 rebound data is critical and often glossed over: Participants who stopped semaglutide after 68 weeks regained an average of 11.6% within a year. Translation: this is a chronic treatment, not a course of antibiotics. (Something the "just take semaglutide for a few months" crowd conveniently ignores.)
SELECT Trial (Cardiovascular Risk Reduction)
SELECT was the trial that proved semaglutide's benefits extend far beyond weight loss:
| Outcome | Semaglutide 2.4mg | Placebo | Risk Reduction | P-value |
|---|---|---|---|---|
| 3-point MACE | 6.5% | 8.0% | 20% | <0.001 |
| Cardiovascular death | 2.5% | 3.0% | 17% | 0.12 |
| Non-fatal MI | 3.4% | 4.0% | 15% | 0.04 |
| Non-fatal stroke | 1.9% | 2.7% | 30% | 0.008 |
| All-cause mortality | 4.3% | 5.1% | 15% | 0.04 |
Source: Lincoff et al., N Engl J Med 2023;389:2221-2232 (SELECT)
Here's the truth most guides bury: SELECT enrolled 17,604 participants with overweight/obesity and established cardiovascular disease but without diabetes. This was the first proof that GLP-1 receptor agonism protects the heart independent of glucose control.
The 30% stroke reduction is particularly striking. If this were a statin trial, it would be front-page news for months.
Ongoing Research: Where Semaglutide Is Heading
| Indication | Trial | Phase | Status |
|---|---|---|---|
| NASH/MASH | ESSENCE | Phase 3 | FDA Accelerated Approval granted Aug 2025 |
| Alzheimer's Disease | EVOKE / EVOKE Plus | Phase 3 | Results expected 2025-26 |
| Post-MI CV risk | SOUL | Phase 3 | Recruiting |
| Diabetic foot ulcers | STRIDE | Phase 3 | Active |
The MASH approval in August 2025 is a big deal. Semaglutide became the first GLP-1 agonist approved for liver disease. The ESSENCE trial showed significant fibrosis improvement in adults with moderate-to-advanced liver fibrosis. For researchers studying metabolic liver disease, this opens an entirely new application space.
EVOKE (Alzheimer's) is the long shot. If semaglutide shows neuroprotective effects in a Phase 3 Alzheimer's trial, the implications for GLP-1 biology research are enormous. Early data suggests the compound may reduce neuroinflammation and amyloid plaque accumulation, but we're waiting on definitive results.
Head-to-Head: Semaglutide vs Tirzepatide vs Liraglutide
Understanding how semaglutide compares to other incretin-based therapies is crucial for research design.
Semaglutide vs Tirzepatide
Tirzepatide represents the next generation as a dual GIP/GLP-1 receptor agonist. Head-to-head data from SURPASS-2 showed:
| Parameter | Semaglutide 1mg | Tirzepatide 5mg | Tirzepatide 10mg | Tirzepatide 15mg |
|---|---|---|---|---|
| HbA1c reduction | 1.86% | 2.01% | 2.24% | 2.58% |
| Weight loss | 6.7 kg | 7.6 kg | 9.3 kg | 11.2 kg |
The SURMOUNT-5 head-to-head (2025) sealed it. This was the first direct comparison of semaglutide 2.4mg vs tirzepatide 15mg for weight management:

SURMOUNT-5 showed tirzepatide nearly doubled the rate of patients achieving 15%+ weight loss (50% vs 23%). But semaglutide still has broader approved indications (diabetes, weight, cardiovascular, MASH) and 5+ more years of safety data.
| Parameter | Semaglutide 2.4mg | Tirzepatide 15mg | Difference |
|---|---|---|---|
| Mean weight loss | 12.0% | 17.8% | +5.8% tirzepatide |
| >=5% weight loss | 71% | 89% | +18% tirzepatide |
| >=15% weight loss | 23% | 50% | +27% tirzepatide |
| >=20% weight loss | 8% | 28% | +20% tirzepatide |
Source: Jastreboff et al., NEJM 2025 (SURMOUNT-5)
Bottom line: Tirzepatide's dual agonism produces superior metabolic effects across the board. But semaglutide has 5+ more years of safety data and a broader approved indication set (diabetes, weight, cardiovascular, MASH). If your research focuses on pure GLP-1 mechanism, semaglutide is the cleaner tool. If you're studying incretin synergy, tirzepatide is the compound.
Key difference: Semaglutide offers pure GLP-1 mechanism with extensive safety data, while tirzepatide provides potentially superior metabolic effects through dual agonism but with less long-term data.
Semaglutide vs Liraglutide
| Feature | Semaglutide | Liraglutide |
|---|---|---|
| Structure | Modified GLP-1 (94% homology) | Acylated GLP-1 (97% homology) |
| Half-life | ~7 days (weekly) | ~13 hours (daily) |
| HbA1c reduction | 1.4-1.8% | 1.0-1.5% |
| Weight loss | 10-15% | 8-10% |
SUSTAIN 10 demonstrated semaglutide's superiority over liraglutide in direct comparison. The weekly vs daily dosing alone is a practical differentiator for research protocols.
The Pipeline Threat: Retatrutide
Here's where it gets interesting for forward-looking researchers.
Retatrutide is a triple agonist: it hits GLP-1, GIP, and glucagon receptors simultaneously. That third receptor activation is what makes it different from everything else in the pipeline.
Phase 2 data (48 weeks):
| Dose | Weight Loss |
|---|---|
| 1mg | 8.7% |
| 4mg | 16.9% |
| 8mg | 22.8% |
| 12mg | 24.2% |
Read that 12mg number again. 24.2% body weight loss in 48 weeks. That's nearly double what semaglutide achieves in 68 weeks.
What the glucagon receptor activation adds: Counterintuitively, activating a receptor that raises blood sugar (glucagon's known function) appears to increase energy expenditure and fat oxidation when combined with GLP-1 and GIP agonism. The net metabolic effect is synergistic, not antagonistic.
Phase 3 trials are ongoing with results expected 2026-27. If confirmed, retatrutide would become the new gold standard for incretin-based weight management research.
For researchers building programs now: Consider designing protocols that can accommodate retatrutide data when it arrives. The receptor binding assays and cAMP accumulation assays you'd run for semaglutide translate directly, you'd just need additional glucagon receptor readouts.
Research Applications

Semaglutide's research footprint extends far beyond weight loss. The five main fields span from established metabolic studies to emerging applications in kidney protection (FLOW trial), Alzheimer's (EVOKE), and addiction research.
Current Laboratory Studies
Metabolic Research:
- Islet cell function and survival studies
- Glucose homeostasis mechanism investigations
- Insulin secretion dynamics analysis
- Beta cell regeneration research
- Adipose tissue browning and thermogenesis
Neuroscience:
- Neuroprotection in diabetes models
- Cognitive function in metabolic disease
- Gut-brain axis signaling studies
- Reward pathway modulation research
- Neuroinflammation reduction (EVOKE trial mechanism hypothesis)
Cardiovascular Biology:
- Endothelial function assessment
- Atherosclerosis progression models
- Cardiomyocyte protection studies
- Inflammatory pathway modulation
- Plaque stabilization research
Hepatology:
- MASH/NASH fibrosis models
- Hepatic steatosis investigations
- Liver inflammation pathway studies
- Fibrosis regression mechanisms
Emerging Areas:
- Addiction and substance use disorders (GLP-1R in reward pathways)
- Polycystic ovary syndrome (PCOS) metabolic management
- Sleep apnea improvement secondary to weight loss
- Kidney protection in diabetic nephropathy (FLOW trial)
- Comparative incretin studies: semaglutide vs liraglutide vs tirzepatide side by side
- Receptor selectivity mapping: pure GLP-1 vs dual GIP/GLP-1 vs triple agonism
Pro Tip: The FLOW trial (2024) showed semaglutide reduced kidney disease progression by 24% in type 2 diabetes patients. If you're designing a research program with broad metabolic scope, the kidney angle is underserved and the clinical data is strong.
In-Vitro Protocol Reference
Receptor Binding Assays:
- Radioligand displacement using [I-125]GLP-1(7-36)NH2
- CHO cells expressing human GLP-1 receptor
- Typical IC50: ~0.1-0.5 nM
cAMP Accumulation:
- Cell lines: INS-1, MIN6, CHO-GLP1R
- EC50 typically 0.1-0.3 nM
Pro Tip: If you're running binding assays and want to compare semaglutide against tirzepatide or retatrutide, set up parallel plates with the same cell passage number. The GLP-1 receptor binding data is directly comparable, but tirzepatide will also show GIP receptor affinity and retatrutide will show glucagon receptor affinity. Having all three on the same plate run eliminates batch-to-batch variability.
Sourcing for Research
If you're buying semaglutide for research, not all sources are equal. Look for vendors who provide Certificates of Analysis (COAs) showing purity of 98% or higher, and mass spectrometry data confirming the molecular structure. Reputable vendors test every batch. The cheapest option often isn't the best when research accuracy matters.
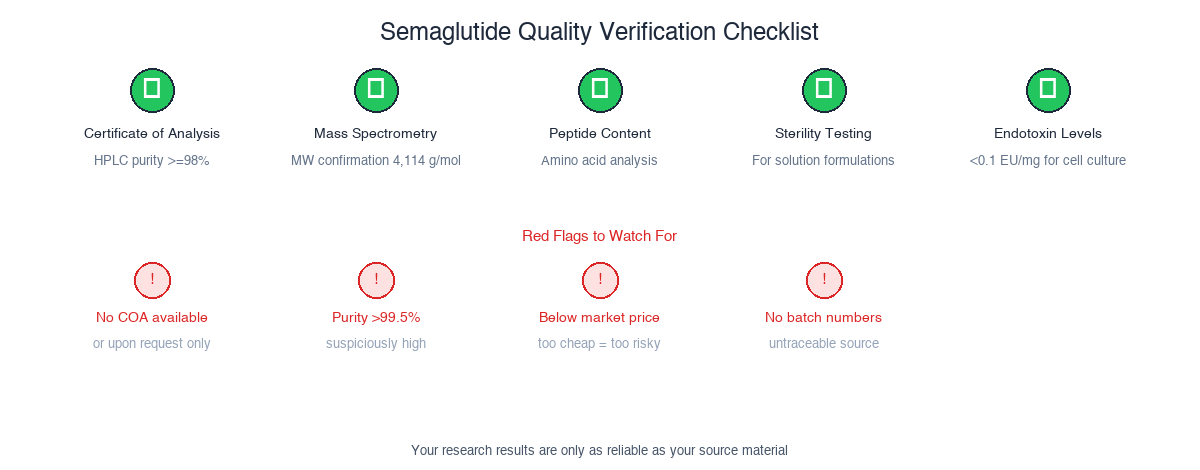
The five non-negotiable checks for any research-grade semaglutide purchase. If a vendor can't provide all five, walk away. The red flags section covers the most common warning signs of low-quality or counterfeit product.
When sourcing semaglutide for laboratory applications, researchers should prioritize quality verification and vendor reliability.
Here's the truth about the research peptide market: quality varies wildly. Some vendors test every batch and publish COAs. Others repackage generic Chinese synthesis without verification. Your research results are only as reliable as your source material.
What to Look for in a Research Vendor
Non-negotiables:
- Third-party Certificate of Analysis (COA) with every batch
- HPLC purity >=98%
- Mass spectrometry confirmation matching expected molecular weight
- Batch-specific lot numbers traceable to production
Nice to have:
- Amino acid analysis for sequence confirmation
- Endotoxin testing for cell culture applications
- Stability data for their specific formulation
- Responsive customer support for technical questions
Red flags:
- No COA available or "upon request only"
- Purity claims >99.5% (suspiciously high for peptides)
- Prices dramatically below market average
- No batch numbers or production dates
Quality Verification Checklist
Before procurement, verify:
- Certificate of Analysis (COA): >=98% purity via HPLC
- Mass Spectrometry: Molecular ion confirmation (4,113.64 g/mol)
- Peptide Content: Amino acid analysis verification
- Sterility Testing: For solution formulations
- Endotoxin Levels: <0.1 EU/mg for cell culture
Storage and Handling
Lyophilized Peptide:
- Storage: -20C (short-term), -80C (long-term)
- Stability: 2-3 years properly stored
- Protect from light and moisture
Reconstituted Solution:
- Use sterile bacteriostatic water
- Storage: 4C for 2-4 weeks
- Avoid repeated freeze-thaw cycles
- Typical concentration: 1-10 mg/mL
Where to Buy Semaglutide for Research
Featured verified vendors that list semaglutide in the directory (up to five, randomized per visit; availability can change):
For the full screened list, see the verified vendor directory.
Conclusion
Semaglutide represents a milestone in peptide therapeutics, demonstrating that structural modifications to native hormones can yield compounds with dramatically improved pharmacokinetics and therapeutic efficacy. For researchers, it offers a well-characterized tool for investigating GLP-1 receptor biology, metabolic regulation, and the expanding therapeutic applications of incretin-based therapies.
The extensive clinical dataset, spanning over 80,000 trial participants, provides an unprecedented foundation for translational research. As investigation continues into neurodegeneration, MASH, and cardiovascular protection, semaglutide remains a central compound in metabolic peptide research.
But here's the bottom line: semaglutide proved that GLP-1 receptor agonism works at scale. Liraglutide proved daily dosing could work, semaglutide proved weekly dosing could work even better, tirzepatide proved adding GIP improves it, and retatrutide is testing whether adding glucagon completes the picture. If you're serious about incretin biology, you need to understand the full lineage. And semaglutide is where the modern story starts.
Where to Go from Here
If you're starting a GLP-1 research program:
- Begin with semaglutide receptor binding assays to establish your baseline
- Add tirzepatide as a comparator for dual agonism studies
- Design your protocols with retatrutide compatibility in mind
- Source from verified vendors and verify every batch
If you're expanding an existing program:
- The MASH/liver fibrosis angle is underserved and clinically validated
- The neuroprotection/EVOKE data (when it drops) could open an entirely new field
- Kidney protection (FLOW trial) is a differentiator few labs are pursuing
For researchers seeking to incorporate this compound into laboratory protocols, explore our verified vendor directory or browse related peptides in our fat loss research category.
Research Notice: This guide is provided for educational and research purposes only. BestPeptide.info does not condone the misuse of research chemicals. Always consult qualified professionals regarding regulatory compliance, safety protocols, and institutional review board requirements.
References: Key studies cited include Marso et al. (2016) SUSTAIN-6, Wilding et al. (2021) STEP-1, Lincoff et al. (2023) SELECT published in the New England Journal of Medicine, and Jastreboff et al. (2023) retatrutide Phase 2 in NEJM. Full citations available upon request.