Semaglutide vs Tirzepatide: A Research Comparison (With Retatrutide Preview)
Head-to-head comparison of semaglutide vs tirzepatide from SURMOUNT-5 trial data. Mechanism differences, clinical outcomes, side effects, cost, and retatrutide pipeline preview.
Semaglutide vs Tirzepatide: A Research Comparison

If you've landed on this page, you're probably wondering which incretin-based compound actually deserves your attention.
Semaglutide and tirzepatide are today's options. But retatrutide, a triple agonist showing 24% weight loss in early trials, is circling the ring. And the landscape is moving fast.
Is tirzepatide's dual mechanism really that much better? Does semaglutide's longer track record matter? And what happens when a third receptor enters the game?
This guide covers all three. Head-to-head data, mechanism differences, pipeline intel. So you can make your own call based on evidence, not hype.
TL;DR
- Tirzepatide produces ~47% more weight loss than semaglutide in the only head-to-head trial (SURMOUNT-5)
- Semaglutide has proven cardiovascular protection: tirzepatide's is still under investigation
- Both share nearly identical side effect profiles (semaglutide actually has slightly worse nausea rates)
- Tirzepatide's dual GIP/GLP-1 mechanism is a genuine pharmacological advantage, not marketing spin
- Retatrutide (triple GIP/GLP-1/glucagon agonist, phase 2) is showing up to 24% weight loss: potentially leapfrogging both current compounds by 2028
At a Glance
| Parameter | Semaglutide | Tirzepatide | Retatrutide (phase 2) |
|---|---|---|---|
| Drug class | GLP-1 receptor agonist | Dual GIP/GLP-1 receptor agonist | Triple GIP/GLP-1/glucagon agonist |
| Manufacturer | Novo Nordisk | Eli Lilly | Eli Lilly |
| Brand names | Ozempic (T2D), Wegovy (weight), Rybelsus (oral) | Mounjaro (T2D), Zepbound (weight) | Not yet approved |
| Mechanism | Mimics GLP-1 only | Mimics both GLP-1 and GIP | Mimics GLP-1, GIP, and glucagon |
| Administration | Weekly SC injection or daily oral | Weekly SC injection | Weekly SC injection |
| Max weight loss (trials) | ~15% at 68 weeks | ~21% at 72 weeks | ~24% at 48 weeks |
| FDA approved | 2017 (Ozempic), 2021 (Wegovy) | 2022 (Mounjaro), 2023 (Zepbound) | Phase 3 ongoing (est. 2027-2028) |
| CV benefit | Proven (SELECT trial) | Under investigation | Under investigation |
How They Actually Work
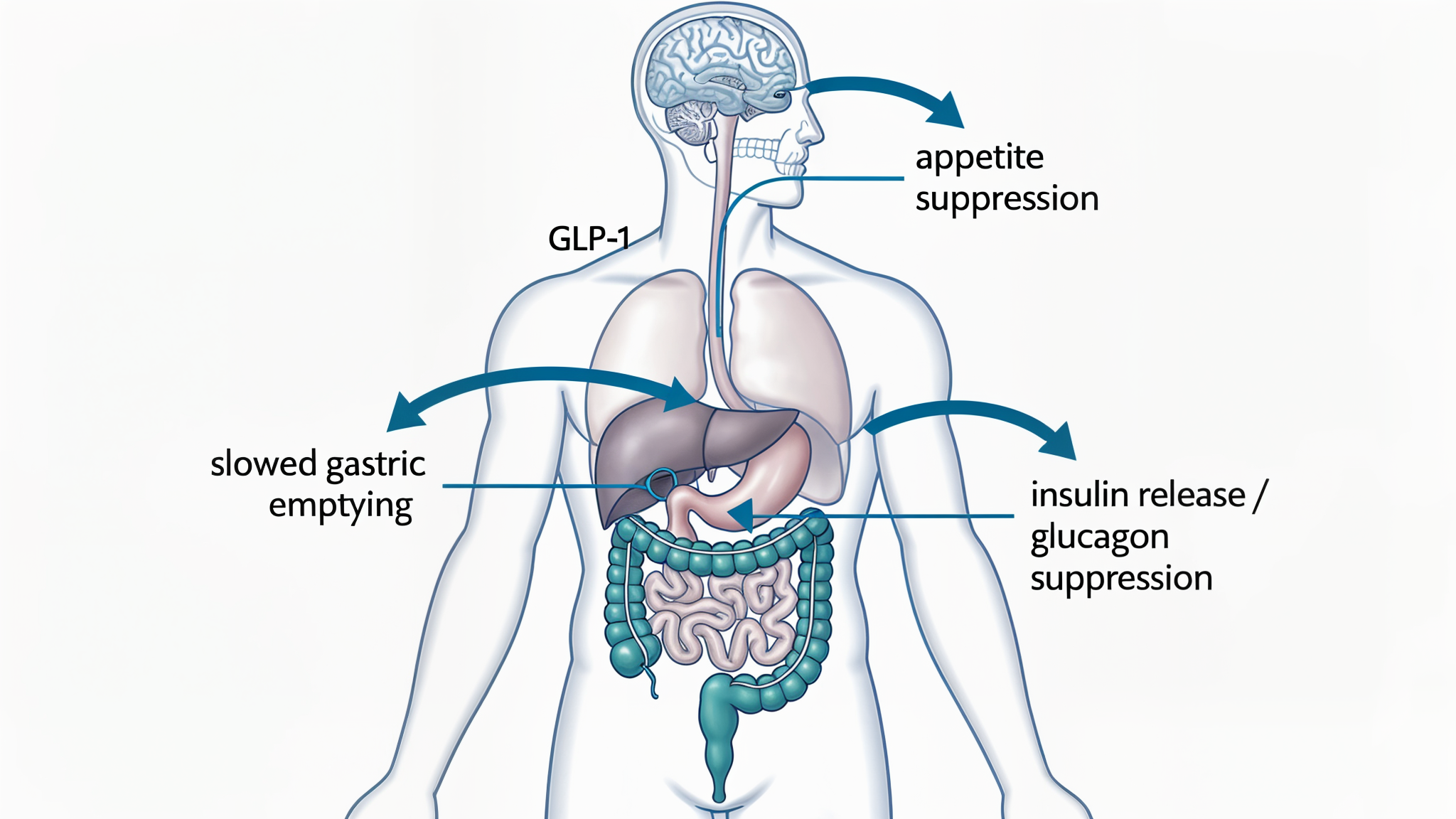
Here's the part most guides get wrong.
They'll say "semaglutide mimics GLP-1" and "tirzepatide mimics GLP-1 and GIP" and then move on like that explains anything. It doesn't.
The mechanism is where the real differences live, and it directly explains why tirzepatide outperforms semaglutide in trials.
Semaglutide: The GLP-1 Specialist
Semaglutide is a selective GLP-1 receptor agonist. It mimics the glucagon-like peptide-1 hormone that your L-cells release after you eat.
What that actually means in practice:
- Gastric emptying slows down: food sits in your stomach longer, so you feel full sooner and stay full longer
- Insulin secretion increases (glucose-dependent, so it won't tank your blood sugar)
- Glucagon release drops: less liver glucose output
- Hypothalamic appetite centers get hit: your brain literally gets a "stop eating" signal
The key structural modification is acylation with a C-18 fatty acid, which extends the half-life to ~7 days. That's what makes weekly dosing possible.
The oral version (Rybelsus) uses an absorption enhancer called SNAC to get semaglutide through the gut wall. But it requires 14 mg daily versus 2.4 mg weekly for injection. Less convenient, but it works.
Tirzepatide: The Dual Agonist
This is where it gets interesting.
Tirzepatide is the first approved compound that hits both the GLP-1 and GIP receptors simultaneously. And the GIP component isn't just a bonus, it changes the equation entirely.
What GIP adds on top of GLP-1:
- Better insulin sensitivity in fat tissue
- Improved lipid metabolism and fat oxidation
- Additional appetite suppression through GIP-specific brain pathways
- Potentially greater lean mass preservation during weight loss (this one is still being studied, but early signals are promising)
Think of it like this: semaglutide turns on the satiety signal. Tirzepatide turns on the satiety signal and optimizes how your body processes what you eat.
That's not a marginal difference. It's a fundamental difference in receptor pharmacology. And it shows up in every trial.
Bottom line: The dual mechanism isn't marketing. It's chemistry. And it's the primary reason tirzepatide consistently outperforms semaglutide across every weight loss threshold.
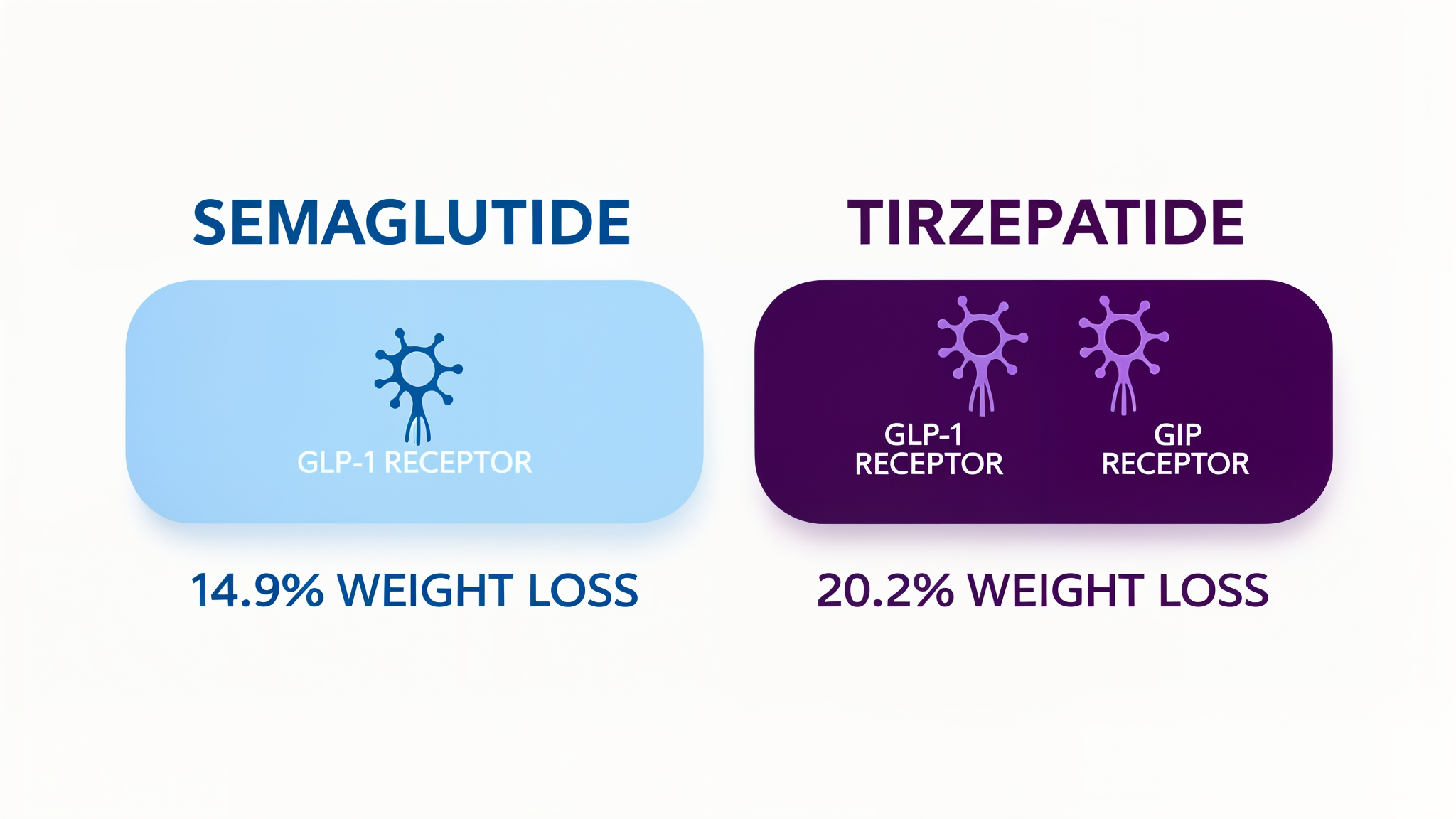
Retatrutide: The Triple Agonist (Phase 2)
If semaglutide is one receptor and tirzepatide is two, retatrutide is the logical next step: three receptors.
Retatrutide is a triple GIP/GLP-1/glucagon receptor agonist developed by Eli Lilly. The same company behind tirzepatide. And the phase 2 data is eye-opening.
What the third receptor (glucagon) adds on top of GLP-1 and GIP:
- Increased energy expenditure: your body burns more calories at rest
- Enhanced fat oxidation: preferential use of fat as fuel
- Hepatic lipid metabolism: better processing of liver fat
- Potential glycogen mobilization: additional metabolic flexibility
The glucagon component is what makes retatrutide different from "just adding more GIP." Glucagon has historically been associated with blood sugar spikes. But when combined with GLP-1 and GIP agonism, the net effect is metabolic optimization rather than hyperglycemia.
Phase 2 results (published in the New England Journal of Medicine):
- At 48 weeks, retatrutide 12 mg produced ~24% weight loss
- That's higher than tirzepatide's 21% at 72 weeks (and retatrutide hit it faster)
- Dose-dependent responses up to 12 mg, with diminishing returns at higher doses
- GI side effects similar to tirzepatide, manageable, not prohibitive
Why this matters for the semaglutide vs tirzepatide debate:
If phase 3 confirms these numbers, retatrutide won't just be "better", it'll be the new benchmark. And Eli Lilly will have both the current champion (tirzepatide) and the next-gen contender.
(Novo Nordisk is countering with CagriSema, a dual amylin/GLP-1 combo showing ~22% in phase 2. The arms race is real.)
Pro Tip: Phase 2 data should be treated as promising, not definitive. Phase 3 trials are larger, longer, and often reveal issues that smaller studies miss. Don't bet the farm on phase 2 numbers. But do keep retatrutide on your radar.
What the Clinical Trials Actually Show
Let's skip the marketing summaries and go straight to the data.
Semaglutide: The STEP Program
The STEP trials (Semaglutide Treatment Effect in People with Obesity) are the backbone of semaglutide's evidence base:
- STEP 1 (n=1,961): 14.9% body weight reduction at 68 weeks (vs 2.4% placebo)
- STEP 2 (n=1,210, T2D patients): 9.6% at 68 weeks
- STEP 3 (n=611, with behavioral therapy): 16.0% at 68 weeks
- STEP 4 (n=803, withdrawal study): stop the drug, regain ~⅔ of the weight
That last one matters. These compounds require ongoing use. (This isn't a "take it for 3 months and you're done" situation.)
But here's the number that actually matters beyond weight loss:
The SELECT trial (n=17,604) showed a 20% reduction in major adverse cardiovascular events: heart attacks, strokes, cardiovascular death, in adults with obesity and existing heart disease.
No other compound in this class has demonstrated that. Not tirzepatide. Not yet.
The STEP program also tracked waist circumference reduction as a secondary endpoint, showing clinically meaningful decreases in central adiposity, which correlates with metabolic syndrome risk.
Tirzepatide: The SURMOUNT Program
The SURMOUNT trials are where tirzepatide flexes:
- SURMOUNT-1 (n=2,539, no T2D): 15.0% (5 mg), 19.5% (10 mg), 20.9% (15 mg) at 72 weeks
- SURMOUNT-2 (n=938, with T2D): 12.8% (10 mg), 14.7% (15 mg) at 72 weeks
- SURMOUNT-3 (n=579, after intensive lifestyle intervention): additional 18.4% (15 mg) at 72 weeks
Notice the dose-response curve. Higher doses = more weight loss, consistently. That's a clean signal.
The One That Matters: SURMOUNT-5 (Head-to-Head)
All the above trials compared each drug to placebo. That's fine for FDA approval, but it doesn't answer the real question: which compound wins when they face each other directly?
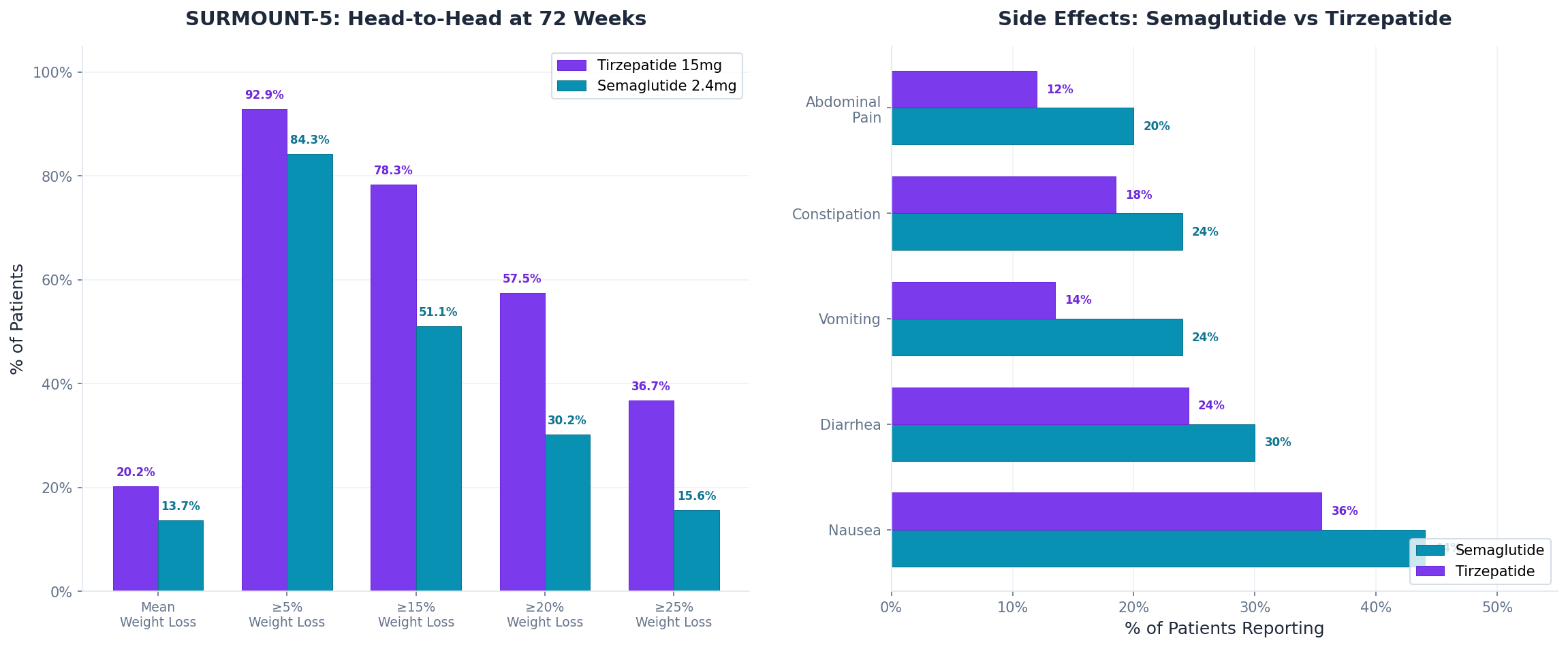
SURMOUNT-5 (n=751) is the only head-to-head trial. Phase 3b, open-label, tirzepatide 15 mg versus semaglutide 2.4 mg, 72 weeks.
The results weren't close:
| Outcome (72 weeks) | Tirzepatide 15 mg | Semaglutide 2.4 mg |
|---|---|---|
| Mean weight loss | 20.2% (50.3 lbs) | 13.7% (33 lbs) |
| ≥5% weight loss | 92.9% | 84.3% |
| ≥15% weight loss | 78.3% | 51.1% |
| ≥20% weight loss | 57.5% | 30.2% |
| ≥25% weight loss | 36.7% | 15.6% |
Read that last row again.
36.7% of tirzepatide users lost 25%+ of their body weight. Only 15.6% of semaglutide users hit that threshold.
The gap doesn't just exist, it widens at higher thresholds. Tirzepatide isn't marginally better. It's substantially, consistently, and clinically significantly superior for weight loss.
Pro Tip: If your primary research endpoint is weight reduction, tirzepatide is the stronger candidate based on current evidence. If cardiovascular protection is the priority, semaglutide remains the only one with trial data.
Side Effects: What to Actually Expect
Here's something that surprises most people:
Semaglutide has worse GI side effects than tirzepatide.
I know. Tirzepatide hits two receptors, so you'd expect more side effects, not fewer. But the data says otherwise:
| Side Effect | Semaglutide | Tirzepatide |
|---|---|---|
| Nausea | 44% | 33-38% |
| Diarrhea | 30% | 23-26% |
| Vomiting | 24% | 12-15% |
| Constipation | 24% | 17-20% |
| Abdominal pain | 20% | 10-14% |
Semaglutide users reported higher rates of nausea, vomiting, diarrhea, and constipation across the board.
(This isn't what you'd expect from a "simpler" compound, but biology is complicated.)
Both carry a boxed warning for thyroid C-cell tumors based on rodent studies. This has not been confirmed in humans, but it's worth noting.
Discontinuation due to adverse events was approximately 7% for both, essentially identical.
Bottom line: Both compounds are well-tolerated by most users. If GI sensitivity is a concern, tirzepatide paradoxically has the better side effect profile.
Dosing Schedules
Both use a gradual escalation protocol: you don't start at full dose. The ramp-up takes 16-20 weeks for both:
Semaglutide (Wegovy):
- Weeks 1-4: 0.25 mg/week
- Weeks 5-8: 0.5 mg/week
- Weeks 9-12: 1.0 mg/week
- Weeks 13-16: 1.7 mg/week
- Week 17+: 2.4 mg/week (maintenance)
Tirzepatide (Zepbound):
- Weeks 1-4: 2.5 mg/week
- Weeks 5-8: 5 mg/week
- Weeks 9-12: 7.5 mg/week
- Weeks 13-16: 10 mg/week
- Then escalate to 12.5 mg or 15 mg as needed
Semaglutide also has an oral option (Rybelsus, 3-14 mg daily), though it's only approved for type 2 diabetes, not weight management. Tirzepatide is injection-only for now.
Pro Tip: The slow ramp-up isn't optional. It's designed to minimize GI side effects. Skipping doses or escalating too fast is the #1 reason people discontinue.
Cost: The Real Numbers
List prices in the US (without insurance):
| Medication | Monthly list price |
|---|---|
| Wegovy (semaglutide 2.4 mg) | ~$1,349 |
| Ozempic (semaglutide) | ~$935-1,029 |
| Zepbound (tirzepatide) | ~$1,060 |
| Mounjaro (tirzepatide) | ~$1,023 |
Here's the reality most guides won't tell you:
- Insurance coverage is a lottery. Medicare Part D began covering anti-obesity meds in 2025, but prior authorization is still the norm.
- Compounded versions exist at $150-500/month through telehealth pharmacies. The FDA has been tightening scrutiny here, worth watching.
- Wegovy is actually more expensive than Zepbound despite being the "weaker" compound.
(The pricing landscape changes fast. Check current rates before making any decisions.)
What's Coming Next: The Pipeline
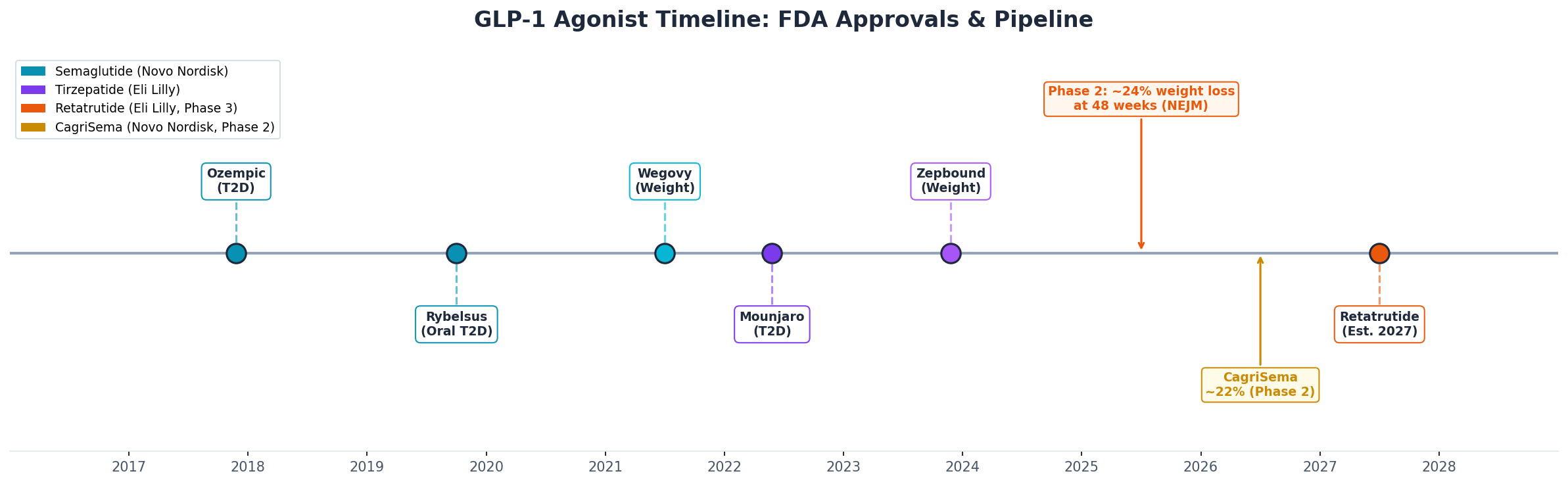
This comparison will look very different in 2-3 years. Several compounds in late-stage trials could reshape the entire landscape:
- Oral semaglutide 50 mg (Novo Nordisk): Phase 3 data showed 15.1% weight loss at 68 weeks: nearly matching injectable semaglutide. If approved, it eliminates the injection barrier entirely.
- CagriSema (cagrilintide + semaglutide): Novo Nordisk's dual amylin/GLP-1 combo showing ~22% weight loss in phase 2. This could leapfrog tirzepatide.
- Orforglipron (Eli Lilly): An oral GLP-1 agonist in phase 3. If it works, it disrupts the injectable-only market from the other direction.
- Retatrutide (Eli Lilly): A triple agonist: GIP/GLP-1/glucagon, showing up to 24% weight loss in phase 2. This is the next generation beyond tirzepatide.
Bottom line: Today's "semaglutide vs tirzepatide" debate is a snapshot. The compounds coming in 2027-2028 will make both of these look like beta versions.
The Verdict
Let's cut through the noise.
If weight loss is your primary endpoint: Tirzepatide wins. Not by a little, by 47% in the head-to-head trial. The dual GIP/GLP-1 mechanism produces superior outcomes across every threshold.
If cardiovascular protection matters: Semaglutide wins. The SELECT trial is the only one in this class that's proven reduced heart attacks and strokes. Until tirzepatide publishes equivalent data, this is semaglutide's unique advantage.
If side effects are a concern: Tirzepatide paradoxically wins here too, lower GI rates across the board despite its dual mechanism.
If cost is the deciding factor: Tirzepatide (Zepbound) is slightly cheaper than semaglutide (Wegovy) at list price. Real-world costs depend entirely on your insurance situation.
If you want an oral option: Semaglutide wins by default. Tirzepatide is injection-only.
If you're thinking long-term: Watch retatrutide. Phase 2 data shows 24% weight loss in 48 weeks, faster and higher than tirzepatide. If phase 3 confirms it, retatrutide becomes the new standard by 2028. Eli Lilly is essentially competing with itself at this point.
Neither compound is "better" in every dimension. But if forced to pick one based purely on the current evidence? Tirzepatide has the stronger clinical profile for weight loss, and the gap is widening with every new trial.
--
This article is for research and educational purposes only. It does not constitute medical advice. Consult a qualified healthcare provider before making any decisions about peptide research or clinical applications.
--
Related Resources:
- Semaglutide Research Guide: Full compound profile, trial data, mechanism of action
- Tirzepatide Research Guide: Complete clinical profile and research findings
- Peptide Vendor Directory: Verified research peptide suppliers
- Peptide Calculator: Reconstitution and dosing calculation tools
- BPC-157 Research Guide: Healing peptide comparison
- Peptide Encyclopedia: Complete directory of all researched compounds
- What Are Peptides?: Introduction to peptide science
- Retatrutide Research Guide: The next-generation triple agonist (coming soon)
- Peptide Research Sources: Where to find clinical-grade research materials
Frequently asked questions
Is tirzepatide stronger than semaglutide?
Yes. In the head-to-head SURMOUNT-5 trial, tirzepatide produced 20.2% weight loss versus 13.7% with semaglutide at 72 weeks, approximately 47% more effective. This is attributed to tirzepatide's dual GIP/GLP-1 receptor mechanism.
Can you switch between semaglutide and tirzepatide?
Yes, under clinical supervision. Dose adjustment and a washout period are typically recommended to minimize overlapping gastrointestinal effects. Always consult a qualified healthcare provider before switching.
Which has fewer side effects?
Tirzepatide. Despite having a dual mechanism, clinical trials showed lower rates of nausea (33-38% vs 44%), vomiting (12-15% vs 24%), and diarrhea (23-26% vs 30%) compared to semaglutide.
Does semaglutide or tirzepatide cause more muscle loss?
Both can reduce lean body mass as part of overall weight loss. Some data suggests tirzepatide may preserve slightly more lean mass, but the primary modifiable factors are protein intake and resistance training, regardless of which compound is used.
Is tirzepatide more expensive than semaglutide?
Actually, no. Zepbound (tirzepatide) costs approximately $1,060/month at list price, while Wegovy (semaglutide) costs approximately $1,349/month. Out-of-pocket costs vary significantly based on insurance and compounding pharmacy access.
What is retatrutide and when will it be available?
Retatrutide is Eli Lilly's triple GIP/GLP-1/glucagon agonist, currently in phase 3 trials. Phase 2 data showed approximately 24% weight loss at 48 weeks, faster and higher than tirzepatide. If phase 3 results confirm these numbers, approval could come as early as 2027-2028. It is not yet available by prescription or for clinical use.
Will retatrutide replace tirzepatide?
Potentially, but not immediately. Even if approved in 2028, tirzepatide will likely remain the standard of care for years while retatrutide builds its evidence base, gains insurance coverage, and scales manufacturing. Think of retatrutide as the "what's next", not the "what's now."
What is CagriSema?
CagriSema is Novo Nordisk's answer to retatrutide, a dual amylin/GLP-1 combination showing ~22% weight loss in phase 2. It pairs cagrilintide (an amylin analog) with semaglutide. If approved, it would be the first non-incretin-based competitor to the GLP-1/GIP class.